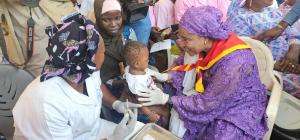
Mali leads the way with innovative hybrid malaria vaccine approach
On World Malaria Day, Mali implements a groundbreaking hybrid vaccination strategy to combat the disease.
The West African nation becomes the 20th African country to introduce malaria vaccines and the first globally to adopt this seasonal dosing approach.
In 2023, Mali accounted for 3.1% of global malaria cases (8.15 million) and 2.4% of global malaria deaths (14,328), ranking among the 11 countries with the heaviest malaria burden worldwide. The hybrid approach will deliver the first three doses according to age, with the fourth and fifth doses administered seasonally before the peak transmission period.
Bamako, Mali — The Ministry of Health and Social Development of Mali, in partnership with Gavi, UNICEF and the World Health Organization (WHO), has launched a pioneering hybrid vaccination strategy targeting children aged 5 to 36 months. This initiative makes Mali the 20th African country to introduce malaria vaccines and the first in the world to implement this innovative dosing schedule.
The hybrid approach combines routine vaccination with seasonal top-ups. Children receive three initial doses throughout the year based on age, followed by two additional doses in May or June before the high-transmission season. This strategy aligns peak vaccine protection with periods of greatest risk, maximizing effectiveness. Data from Mali demonstrates this approach significantly enhances malaria prevention.
The R21/Matrix-M vaccine will initially be deployed in 19 priority districts across five regions: Kayes, Koulikoro, Mopti, Ségou and Sikasso. Mali has received 927,800 doses for this rollout.
Mali’s malaria challenge
The WHO’s 2024 World Malaria Report highlights Mali’s severe burden, with the country accounting for 3.1% of global cases and 2.4% of global deaths in 2023. Among the 11 nations with the highest malaria burden, Mali saw a 1.4 million case increase between 2019 and 2023. The African Region bears 94% of global cases and 95% of deaths.
During the launch ceremony, Mali’s Minister of Health and Social Development, Colonel Assa Badiallo Touré, expressed gratitude to Gavi, the Global Fund, WHO and UNICEF for their unwavering support in introducing the malaria vaccine into the country’s Expanded Programme on Immunization (EPI).
“This vaccine introduction represents monumental collective effort,” said Colonel Touré. “Our researchers’ contributions during clinical trials were instrumental in WHO’s recommendations for RTS,S and R21 vaccines. While we celebrate this milestone, we recognize the challenge ahead in scaling up this intervention to reduce malaria’s devastating impact on our population.”
Global partnership and funding
Gavi coordinates the global malaria vaccination program, partnering with countries and organizations to fund vaccine procurement, transportation and deployment. Through its unique co-financing model, countries gradually increase their financial contributions to vaccination programs. The future of malaria vaccination faces a critical juncture as Gavi seeks funding for its 2026-2030 strategic period.
Dr. Sania Nishtar, Gavi CEO, emphasized the importance of sustained investment: “Mali’s commitment to protecting families from malaria’s devastating effects is commendable. With 20 countries now deploying malaria vaccines and over 24 million doses delivered, consistent funding remains essential to ensure this life-saving tool reaches those who need it most.”
UNICEF’s role in vaccine delivery
UNICEF plays a crucial role in vaccine distribution and demand generation. Dr. Pierre Ngom, UNICEF Representative in Mali, noted: “After 35 years of dedicated research, the malaria vaccine finally provides a powerful new tool to protect Malian children. While revolutionary, this vaccine is not a standalone solution. We’re supporting community mobilization efforts to complement existing prevention measures.”
UNICEF’s youth volunteers leverage digital tools like U-Report chatbots to combat misinformation and promote vaccination in communities.
WHO’s vaccine validation
The WHO coordinated pilot evaluations of the RTS,S/AS01 vaccine in Ghana, Kenya and Malawi through the Malaria Vaccine Implementation Programme (MVIP), co-funded by Gavi, the Global Fund and UNITAID. Between 2019-2023, over two million children received the vaccine, resulting in a 13% reduction in child mortality. These results supported WHO’s recommendation and prequalification of both malaria vaccines.
Dr. Patrick Kabore, WHO Representative in Mali, stated: “The malaria vaccine represents one of public health’s most significant recent advances, providing critical protection for children in our ongoing fight against this deadly disease.”
The vaccine complements Mali’s existing malaria prevention measures, including insecticide-treated nets, seasonal malaria chemoprevention, intermittent preventive treatment in pregnancy and indoor residual spraying.
Global malaria vaccine rollout progress
Mali’s vaccine introduction coincides with Uganda’s recent large-scale deployment, marking another milestone in Africa’s fight against malaria. Since 2023, over 24 million vaccine doses have been delivered across the continent, with countries progressively expanding their programs.
The 20 African countries introducing vaccines now represent over 70% of the global malaria burden. Early results from Cameroon demonstrate promising outcomes, with additional countries planning introductions this year. By the end of 2025, an estimated 13 million more African children should be protected by malaria vaccines.
How the hybrid vaccination strategy works
Mali’s seasonal transmission pattern means most cases occur between July and December. The hybrid approach ensures children receive:
- The first three doses throughout the year based on age
- The fourth and fifth doses seasonally in May or June, before peak transmission
Evidence shows this alignment of vaccine effectiveness with transmission risk periods significantly enhances impact.
Why children are the primary target
Children under five face the highest risk of malaria-related death, accounting for over 75% of global malaria deaths. Unlike adults, young children lack partial immunity developed through years of exposure, making them particularly vulnerable.
Vaccine safety and efficacy
The WHO-prequalified RTS,S/AS01 and R21/Matrix-M vaccines are both safe and effective for preventing malaria in children.
- Phase 3 clinical trials showed both vaccines reduced malaria cases by over 50% in the first year after vaccination, a critical period of high risk
- A fourth dose in the second year extended protection
- Both vaccines reduce malaria cases by about 75% when administered seasonally in high-transmission areas
- These vaccines target Plasmodium falciparum, the deadliest and most prevalent malaria parasite in Africa
Adapting vaccination programs
Administering four or five doses requires systematic approaches to reach at-risk populations. Countries are implementing tailored strategies:
- Nigeria: Began phased rollout in Kebbi and Bayelsa states in December 2024, targeting areas with high malaria prevalence
- Chad: Integrated malaria vaccines into a triple vaccination campaign against malaria, pneumonia and diarrhea
- Sudan and Democratic Republic of Congo: Integrated rollouts into broader response plans